 
Basically, ISO 13485 is like a quality management system for organizations involved in design, production, installation, and servicing of medical devices, with some other important requirements for good measure. ISO 13485 is designed to be used by organizations involved in the design, production, installation and servicing of medical devices and related services. The manual is divided into eight sections that correlate to the Quality Management System sections of ISO 13485:2016. 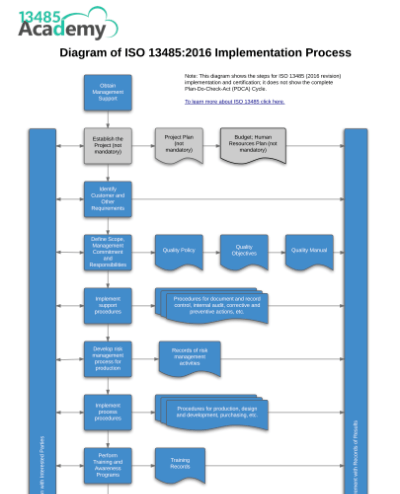
Iso 13485 pdf ISO 13485, Annex A provides some detailed commentary on the changes between the 20 editions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
